
Evaluating Benefits of PD-L1 Image Analysis for the Clinical Setting
Key takeaways Our spatial biology approach using Flagship’s unique analysis technology reduced variability in tissue scoring Compared to manual scoring, ourAI-driven scoring of non-small cell
Analytical Validation of Ki67/CD8 Duplex IHC Assay Using Computational Tissue Analysis (cTA™)
Key takeaways Ki67/CD8 chromogenic duplex IHC positive cell staining was quantified using our cTA™ technology Both Ki67 nuclear and CD8 membrane staining were accurately and
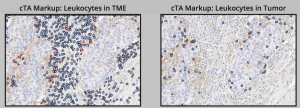
Computational Tissue Analysis-Based Quantification of Tumor-Infiltrating Leukocytes Using Morphometrics in Immunohistochemistry-Stained NSCLC Samples
Abstract Background: Quantification of tumor-infiltrating lymphocytes (TILs) is predictive of patient response to immunotherapy regimens. Because of the role of anti-PD-L1 and anti-PD-1 treatments in
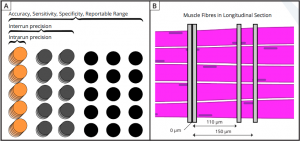
Analytical Validation (Based on CLIA & CLSI Standards) of Utrophin–Laminin α2 (Merosin) and MHCd-Laminin α2 Duplex Immunohistochemical Assays Using Computational Tissue Analysis (cTA™) for Evaluation of Duchenne Muscular Dystrophy Therapeutics
Abstract: Tissue–based assays provide insight for understanding therapeutic response for Duchenne muscular dystrophy (DMD) therapies within the context of intact muscle. This necessitates that at
Computational Alignment of Duplex Immunohistochemically-Stained Muscle Sections in Support of Therapies for Duchenne Muscular Dystrophy
Abstract: Continuous expression of utrophin protein by a utrophin modulator could be a disease modifying treatment for Duchenne muscular dystrophy (DMD) patients regardless of their
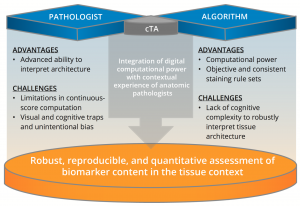
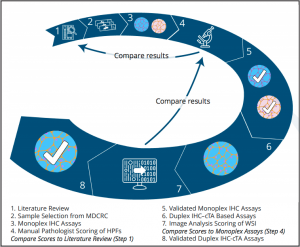
Evaluating Image Analysis Approaches Toward “Harmonization” of PD-L1 Assays
Abstract: The commercial diagnostic landscape for PD-L1 immunohistochemistry (IHC) assays is highly complex. Multiple different companion or complementary diagnostic tests exist for therapeutics targeting the

