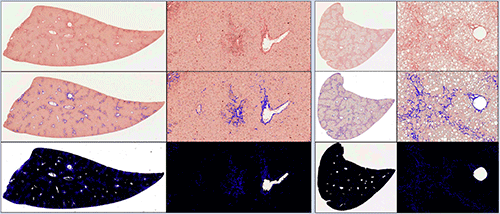
Introduction:
Nonalcoholic steatohepatitis or NASH is a common, often “silent” chronic liver disease. While resembling alcoholic liver disease, it occurs in people who drink little or no alcohol. The lesions most commonly accepted for NASH include steatosis, hepatocyte ballooning degeneration, mild diffuse lobular mixed acute and chronic inflammation, and perivenular, perisinusoidal collagen deposition. Progression of fibrosis may result in bridging septa and cirrhosis, ultimately leading to liver failure. There are no specific therapies for NASH. Current treatment focuses on controlling associated medical conditions, such as diabetes and obesity, and on monitoring for progression. Emerging antifibrotic therapies are aimed at inhibiting the accumulation of fibrogenic cells and/or preventing the deposition of extracellular matrix proteins. Development of a reproducible murine model recapitulating the progressive nature of NASH with accurate and reproducible detection and analysis of fibrosis progression would be a useful tool for studying the natural history, molecular mechanisms and biology of NASH.
Animal models mimicking NASH are in development. A major obstacle in their utilization is the identification and quantification of induced fibrosis and its amelioration due to therapy. Previous attempts to generate such models have reproduced the metabolic and inflammatory aspects of NASH without the development of progressive liver fibrosis. A diet-inducing murine model has resulted in a liver with many clinical features of NASH including hepatic fibrosis. However, fibrosis generally occurs at a low area percentage in tissues, and manual subjective evaluation cannot accurately discern discreet changes at low levels. Quantitative tissue image analysis (tIA) solutions have been developed to accurately and consistently determine percentages of induced fibrosis in various experimental animal models. This data in combination with histomorphologic and molecular data allows a greater utilization of these animals in antifibrotic drug development.